Our solution approaches
 Implementing the requirements of Annex 1 is often complex in practice. Sterile manufacturing processes face challenges such as maintaining contamination‑free airflow, minimizing critical interventions, efficiently monitoring cleanrooms, and ensuring both regulatory compliance and process security. The integration of barrier technologies, automation, and qualified personnel also presents organizational and technical hurdles for many companies.
Implementing the requirements of Annex 1 is often complex in practice. Sterile manufacturing processes face challenges such as maintaining contamination‑free airflow, minimizing critical interventions, efficiently monitoring cleanrooms, and ensuring both regulatory compliance and process security. The integration of barrier technologies, automation, and qualified personnel also presents organizational and technical hurdles for many companies.Our filling and closing systems are specifically designed to address these challenges. With RABS and isolator technologies, optimized First‑Air concepts, fully integrated environmental monitoring solutions, and automated processes, we reduce human interaction to an absolute minimum. At the same time, our systems support QRM and CCS strategies through comprehensive documentation, validated processes, and continuous monitoring.
In this way, we combine compliance, safety, and efficiency: your production not only meets the stringent requirements of Annex 1 but also becomes more robust, more reliable, and future‑proof.
Without sorting bowls. Contamination-free.
The next generation of stopper feeding.
The pharmaceutical industry is facing new challenges due to the stricter requirements of Annex 1. In particular, uninterrupted airflow (First Air) and the full sterilizability of all product-contact parts are putting increasing pressure on traditional stopper feeding systems with sorting bowls. Conventional systems disrupt First Air, carry contamination risks, and make cleaning and autoclaving more difficult.The answer: Our bowl-free feeding solution
With the aseptic bowl-free-feed (BFF), we have developed a future-proof solution that completely eliminates sorting bowls – enabling maximum sterility, simplified processes, and the highest level of automation.
Your advantages at a glance
- Compatibility with all types of stoppers for vials, syringes, and cartridges
- High speed – processes up to 60,000 stoppers per hour for maximum efficiency
- Hygienic design for easy cleaning – magnetic technology enables thorough cleaning
- Reduced manual intervention through robot-assisted systems
- Automatic aseptic setup possible
- Zero-reject principle for maximum output+B6:C6
The BFF solves key challenges in aseptic manufacturing: it improves regulatory compliance, reduces contamination risks, and increases efficiency. By combining robotics with state-of-the-art hygienic design, it sets a new standard for sterile stopper feeding – preparing pharmaceutical filling and closing systems for the requirements of tomorrow.
Continuous Monitoring
 The revised EU GMP Annex 1 sets clear requirements for the continuous monitoring of particles and other critical parameters throughout the entire operation of aseptic processes. In particular, microbiological environmental monitoring must be seamless, reproducible, and carried out without interrupting or impairing production.
The revised EU GMP Annex 1 sets clear requirements for the continuous monitoring of particles and other critical parameters throughout the entire operation of aseptic processes. In particular, microbiological environmental monitoring must be seamless, reproducible, and carried out without interrupting or impairing production.To reliably and efficiently meet these expectations, we offer a fully automated settle plate handling system for the complete processing of sedimentation plates.
Our system enables the integration of a continuous microbiological monitoring program in which the handling of the plates is fully automated. This relieves operating personnel during ongoing production and reduces human interaction in critical areas to an absolute minimum.
Your benefits at a glance
- Optimal First‑Air immersion
- Reliable through automated handling: reduces manual interventions and minimizes contamination risk
- No mix‑up of exposed and unexposed settle plates: clear process logic and unambiguous assignment ensure full traceability
- Suitable for HPAPI processing: safe use even in high‑potency active ingredient environments
Gloveless solutions
The revised Annex 1 significantly increases the requirements for sterility and process security in aseptic manufacturing. Every human intervention is considered a risk – even through isolator gloves. Gloves can fatigue, become damaged, or develop undetected leaks, making them a potential weak point in automated aseptic environments.Gloveless solutions eliminate this risk entirely by removing gloves from the process and enabling fully enclosed, automated operations. This drastically reduces contamination risks while ensuring standardized, reproducible workflows. In combination with robotics, sensor technology, and data‑driven monitoring, gloveless concepts establish a new level of stable, safe asepsis – fully aligned with Annex 1 and the future of sterile manufacturing.
GENEX: The next generation of gloveless filling

GENEX is a fully gloveless process solution specifically designed for aseptic small‑batch manufacturing. It was developed to significantly reduce contamination risks. By relying entirely on robotics, GENEX eliminates all manual interventions in critical process steps such as handling, filling, stoppering, and crimping. This prevents human error and ensures reliable compliance with EU‑GMP Annex 1 and FDA guidelines.
The platform is modular, scalable, and extremely compact – ideal for biologics, vaccines, and ATMPs that require absolute sterility, fast changeovers, and minimal product loss. With the industry moving toward high‑value, sensitive therapies produced in small batches, GENEX delivers the flexibility needed: robotic modules can be quickly programmed for new formats, and the system can be reconfigured without long downtimes. In this way, GENEX combines maximum aseptic safety with high agility.
GENEX Benefits
- Minimized contamination risk thanks to fully robot‑controlled, gloveless isolator operation
- Maximum flexibility through robotic, GMP‑compliant format changes
- Ideal for clinical studies and small batches across a wide range of product types and fill volumes
- Accelerated regulatory approvals enabled by reproducible, validatable, and standardized processes
Hygienic design in the context of Annex 1
 With the revision of EU GMP Annex 1, the requirements for hygienic design in sterile filling equipment have increased significantly. The goal is to eliminate every potential source of contamination and ensure the highest level of cleanability and bio‑decontamination.
With the revision of EU GMP Annex 1, the requirements for hygienic design in sterile filling equipment have increased significantly. The goal is to eliminate every potential source of contamination and ensure the highest level of cleanability and bio‑decontamination.A central approach is the redesign of our object transfer concept. No moving parts are located in “grey” or undefined zones. All functional components are assigned to clearly defined, controlled areas – an essential prerequisite for compliance, risk reduction, and audit readiness.
Consistent hygienic design also plays a key role in maintaining First Air. Streamlined geometries, reduced component heights, and the avoidance of unnecessary structures ensure that critical zones – especially in the filling area – remain continuously supplied with aseptic airflow and are not obstructed by mechanical components or disturbed by turbulence.
The result is a holistic system concept with optimal cleanability, efficient bio‑decontamination (e.g., VHP), shortened downtime, and maximum product safety.
Annex 1 compliance through CFD studies
EU‑GMP Annex 1 requires demonstrable product protection during all critical steps of the aseptic filling and closing process. A key element is the maintenance of First Air conditions in the product‑proximate area. CFD studies (Computational Fluid Dynamics) provide a robust, simulation‑based method to demonstrate this.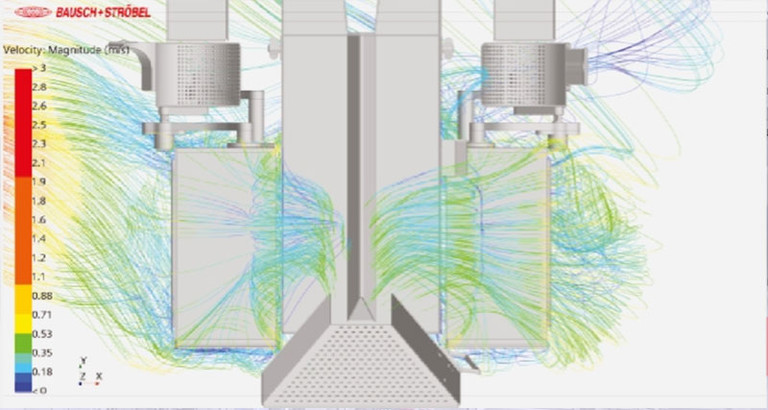
Using CFD analyses, airflow around filling needles, stoppering processes, and crimping stations is simulated and visualized in detail. This makes it possible to evaluate early on whether the required, unobstructed airflow toward the product is maintained – and how machine design, barrier components, or pressure zones may influence product protection.
One major advantage of CFD studies is their ability to be applied very early in the project lifecycle. During the design, planning, or retrofit phase, critical zones can be identified and optimized long before a machine goes into operation. The resulting data serves as transparent, technically sound documentation and supports manufacturers in demonstrating Annex‑1‑compliant First‑Air performance in a clear and verifiable manner to regulatory authorities.
Achieving Annex 1 compliance with Omnia
EU‑GMP Annex 1 requires comprehensive and traceable control of aseptic filling and closing processes throughout the entire lifecycle of a production line. With Omnia, we provide a digital solution that helps manufacturers implement these requirements in a structured and data‑driven way.
Omnia serves as a central platform for collecting, analyzing, and documenting all process‑relevant information along filling and closing lines. This includes process states, interventions, alarms, as well as selected environmental and operational parameters in product‑proximate areas. The consistent consolidation of this data creates transparency about the actual operating condition of the line and enables an objective assessment of deviations and risks.
Through standardized evaluations, clear visualizations, and audit‑proof documentation, Omnia supports well‑founded decision‑making during routine operation and process changes. At the same time, the structured data foundation simplifies the preparation and execution of audits and inspections. In this way, Omnia makes a significant contribution to ensuring sustainable compliance with Annex 1 requirements for aseptic filling and closing processes.
PUPSIT – Safety before production start
PUPSIT (Pre‑Use Post‑Sterilization Integrity Test) refers to the integrity test performed on the sterilized filter immediately before the start of production. This ensures that the final product filter is tested in exactly the same condition in which it will be used for aseptic filling.Transport, installation, and sterilization can cause damage to the filter membrane. In the past, integrity testing was often performed only at the end of production – accepting the risk that potential defects might be detected only after filling. Today it is well known that the product itself can mask even very small defects and falsify the test results.
PUPSIT is now considered an essential element of a compliant manufacturing process – ideally performed as close as possible to the point of use to ensure maximum product safety.
We integrate the complete product‑filter setup directly into the filling machine design – close to the point of use, either inside or outside the isolator. Whether single‑use assemblies, SOP‑ or CIP/SIP‑based systems, single or double filtration: the configuration is designed to be process‑safe and application‑specific.
Automated filter wetting using product, WFI, or buffer solution, combined with established test methods such as Forward Flow, Bubble Point, or Water Intrusion Test, ensures stable and reliable operation immediately before production start – including full documentation in the batch report.
Integrity‑test devices from all major manufacturers are integrated into the machine control system; test results and critical process parameters such as pressure and flow are continuously recorded. In addition, we optimize the filtration design to minimize product loss – both in new equipment and in retrofit projects, independent of filter type or supplier.
Upgrading existing lines for safe and future‑proof operation
The requirements of the revised EU‑GMP Annex 1 present many pharmaceutical manufacturers with the same challenge: how can existing production lines be adapted to meet the tightened expectations for contamination control, risk management, and barrier technologies – without compromising ongoing operations? We support you exactly at this point. Our service helps you systematically evaluate your existing equipment for Annex‑1 compliance and clearly define the measures needed to achieve it. Together, we analyze the current condition of the line, identify potential risks, and outline concrete optimization and retrofit options – technically sound, practical, and independent of the original equipment manufacturer.
We support you exactly at this point. Our service helps you systematically evaluate your existing equipment for Annex‑1 compliance and clearly define the measures needed to achieve it. Together, we analyze the current condition of the line, identify potential risks, and outline concrete optimization and retrofit options – technically sound, practical, and independent of the original equipment manufacturer.Whether minor adjustments, targeted modernizations, or comprehensive retrofit solutions: we advise you transparently on all available options and support you from the initial assessment through to successful implementation. This gives you a solid basis for decision‑making and a clear action plan to reliably bring your equipment up to current regulatory standards.
With our support, you establish the foundation for a robust Contamination Control Strategy and long‑term Annex‑1 compliance – efficient, safe, and future‑ready.
